Study Designs
More EBP
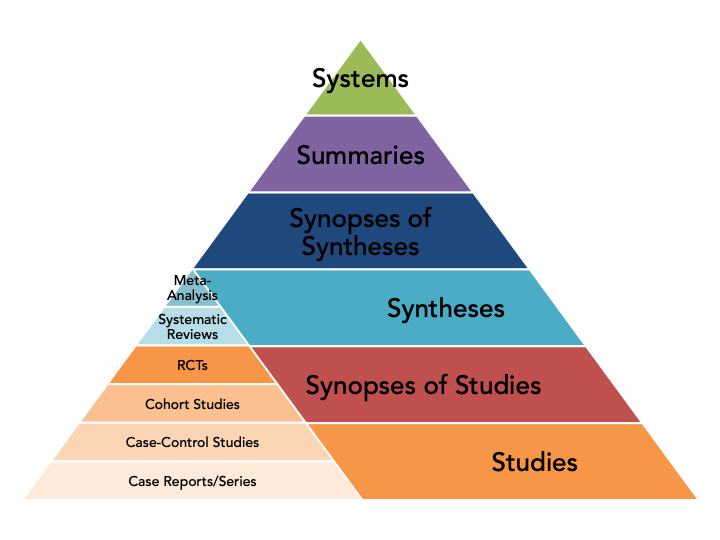
Evidence Pyramid
This evidence pyramid integrates the 6S model (the large pyramid) with the hierarchy of study types (the small inset pyramid). As the pyramid progresses upwards the level of evidence increases. For definitions of each type of evidence continue reading below.
Evidence Type Definitions
Definitions were taken from the Centre for Evidence-Based Medicine's Glossary, Accessing Preappraised Evidence: Fine Tuning the 5S Model into a 6S Model, and from the MeSH Database.
Systems
An electronic medical record that is able to link updated, evidence-based information related to a patient's clinical information. Also known as an integrated computerized decision support system. Very few exist at this time.
Summary
These include topic summaries and clinical practice guidelines that integrate evidence-based information about specific clinical problems with regular updating.
Synopses of Syntheses
A synthesis would be a systematic review (see definition below). A synopsis of that synthesis would be an expert/authoritative appraisal or critique of a high quality systematic review that provides sufficient information to support clinical action.
Syntheses
A summary of the medical literature that uses explicit methods to perform a comprehensive literature search and critical appraisal of individual studies. There are two categories of synthesis.
-
Systematic Review: The application of strategies that limit bias in the assembly, critical appraisal, and synthesis of all relevant studies on a specific topic. Systematic reviews focus on peer-reviewed publications about a specific health problem and use rigorous, standardized methods for selecting and assessing articles. A systematic review may or may not include a meta-analysis.
-
Meta-Analysis: A quantitative integration of data from individual studies using statistical techniques.
Synopses of Single Studies
An expert/authoritative appraisal or critique of an individual study that provides sufficient information to support clinical action.
Studies
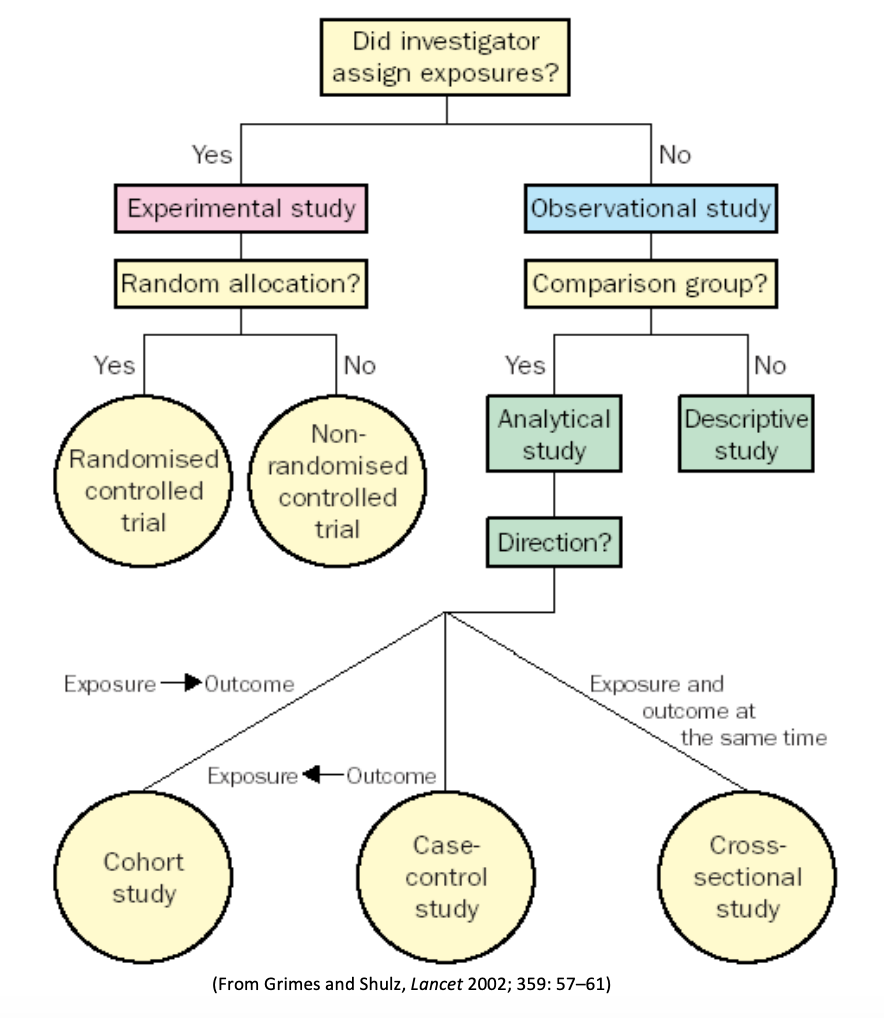
Individual studies, including both experimental and observational studies.
-
Randomized Controlled Trial: An experimental study in which subjects in a population are randomly allocated into groups, usually called intervention and control groups, to receive or not receive an experimental preventive or therapeutic procedure, maneuver, or intervention. The results are assessed by rigorous comparison of rates of disease, death, recovery, or other appropriate outcome in the study and control groups.
-
Cohort Studies: An observational study in which subsets of a defined population can be identified who are, have been, or in the future may be exposed or not exposed, or exposed in different degrees, to a factor or factors hypothesized to influence the probability of occurrence of a given disease or other outcome. The main feature of a cohort study is observation of large numbers over a long period (commonly years) with comparison of incidence rates in groups that differ in exposure levels. Cohort studies can be prospective or retrospective. Learn more about cohort studies with this short video.
-
Case-Control Study: An observational study of persons with the disease (or other outcome variable) of interest and a suitable control (comparison, reference) group of persons without the disease. The relationship of an attribute to the disease is examined by comparing the diseased and nondiseased with regard to how frequently the attribute is present or, if quantitative, the levels of the attribute, in each of the groups. Learn more about case-control studies with this short video.
-
Cross-Sectional Study: An observational study that examines the relationship between diseases (or other health-related characteristics) and other variables of interest as they exist in a defined population at one particular time (ie exposure and outcomes are both measured at the same time). Best for quantifying the prevalence of a disease or risk factor, and for quantifying the accuracy of a diagnostic test.
-
Case Reports/Series: A report on a single patient or series of patients who were given similar treatment. Case reports/series usually contain detailed information about the individual patients. This includes demographic information (for example, age, gender, ethnic origin) and information on diagnosis, treatment, response to treatment, and follow-up after treatment.
Learn More
- Introduction to Study Design, a handout from the Centre for Evidence-Based Medicine
- Study Design 101, a tutorial that breaks down different types of studies and how they relate to each other, designed by the Himmelfarb Health Sciences Library
